 
644 Chromium ( III ) iodide 645 Chromium ( III ) nitrate Formula C1,06 CI20 CIF CIF. The red-violet hydrate is highly soluble in water. 619 Chromium nitride 620 Chromium nitride 621 Chromium phosphide 622. The anhydrous salt forms green crystals and is very soluble in water (in contrast to anhydrous chromium(III) chloride which dissolves very slowly except under special conditions). The chromium centers are bound to six aquo ligands, and the remaining volume of the solid is occupied by three nitrate anions and three molecules of water of crystallization. Chromium(III) nitrate compounds are of a limited importance. Chromium has a 2+ charge and nitrate has a 1- charge, so one chromium ion and two nitrate. Most common is the dark violet hydrated solid, but an anhydrous green form is also known. 3H 2O - betray a simple structure of this material. The preparation of hexammine chromium(III) nitrate, Cr(NH3)6(NO3)3, can be performed by a minimally complicated two-step process involving ammoniation of CrCl3. In chromium(II) nitrate, there is one chromium ion in the formula.The relatively complicated formula - (NO 3) 3 It is common in academic laboratories for the synthesis of chromium coordination complexes. The synonyms, trade name, chemical formula, and identification numbers of chromium and selected salts are reported in Table 4-1. Chromium(III) nitrate compounds are of a limited commercial importance, finding some applications in the dyeing industry. CHEMICAL AND PHYSICAL INFORMATION 4.1 CHEMICAL IDENTITY Information regarding the chemical identity of chromium is located in Table 4-1. Most common is the dark violet hygrosscopic solid. ammonia + sulfuric acid ammonium sulfate 2NH 3 + H. SO 4 2-nitrate: NO 3-hydrogen sulfate: HSO 4-arsenite: AsO 3 3-thiosulfate: S 2 O 3 2-fluoride: F. lead (II) hydroxide lead (II) oxide + water Pb(OH) 2 PbO + H 2O 37. Formula: Name: Formula: zinc: Zn +2: chromium(II) Cr +2: mercury(II) Hg +2: hydronium: H 3 O + ferric: Fe +3: manganese(II) Mn +2: hydrogen: H + strontium: Sr +2: silver: Ag + nitronium: NO 2 + barium: Ba +2: stannic: Sn +4: magnesium: Mg +2. sodium nitrate sodium nitrite + oxygen 2NaNO 3 2NaNO 2 + O 2 36. It is used as an additive in drugs, anti-corrosive pigments, catalysts and polymer manufacture.Except where otherwise noted, data are given for materials in their standard state (at 25 ☌, 100 kPa).Ĭhromium(III) nitrate describes several inorganic compounds consisting of chromium, nitrate and varying amounts of water. copper (II) nitrate + ammonium hydroxide copper (II) hydroxide + ammonium nitrate Cu(NO 3) 2 + 2NH 4OH Cu(OH) 2 + 2NH 4NO 3 35. Chromium(III) phosphate undergoes thermal decomposition at a high temperature of 14500C to yield chromium oxide and phosphorus pentaoxide. Formula: Cr(NO3)3.9H2O Hill system formula: Cr1H18N3O18 CAS registry number: Formula weight: 400.148 Class: complex Colour: green-black. 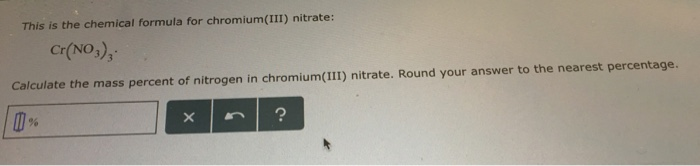
The melting point of CrPO4 stands at a temperature of 1000C however it is a stable compound at normal temperature and pressure. Chromium trinitrate Molecular Formula, CrN3O Molar Mass, 238.008 g/mol InChI. It is insoluble in water but soluble in acids. Chromium nitrate Alternative Names, Chromic nitrate. CrPO4 normally exists as a green crystal, CrPO4•4H2O, or a violet crystal, CrPO4•6H2O, in its hydrated form. In nature, it occurs as the rare mineral eskolaite.Chromium(III) phosphate is an inorganic compound with the chemical formula CrPO4. It is one of the principal oxides of chromium and is used as a pigment. 
It is common in academic laboratories for the synthesis of chromium coordination complexes.Chromium(III) oxide is the inorganic compound of the formula Cr2O3. Chromium trinitrate is an inorganic nitrate salt consisting of chromium and nitrate in which the ratio of chromium (in the +3 oxidation state) to nitrate is 1:3 It is an inorganic nitrate salt and a chromium coordination entity. Chromium(III) nitrate compounds are of a limited importance commercially, finding some applications in the dyeing industry. 
Most common is the dark violet hydrated solid, but an anhydrous green form is also known. Chromium(III) nitrate describes several inorganic compounds consisting of chromium, nitrate and varying amounts of water. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
